Assessment of drug related problems in patients prescribed with antibiotics in a tertiary care hospital
 DOI:
DOI:
https://doi.org/10.33974/gr8sws74
Keywords:
Antibiotics, DRP’S, Cephalosporins, ADR’sAbstract
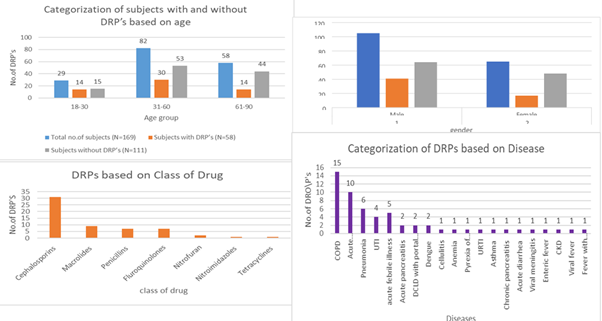
India has one of the greatest burdens of bacterial infections in the world and the crude mortality rate is 417 per 100,000 persons. The present research was conducted with the purpose to evaluate drug-related issues (DRPs) among patients receiving antibiotics in a tertiary care hospital. A potential observational study was carried out in 170 inpatients at the General Medicine Department of Government General Hospital (RIMS) that was conducted in Kadapa between October 2024 and April 2025. The selection of patients was done according to the willingness and inclusion criteria, and the ethical approval was received within the Institutional Human Ethics Committee. Descriptive and inferential statistical analysis that used p-value estimation were carried out with Jamovi v2.6.44.0. DRPs were the most prevalent among the participants aged 31-60 years (30 cases; 51.72). The proportion of males (70.68) was higher than that of females (29.32). The cephalosporins were the class of antibiotics with the greatest number of DRPs (31 cases; 53.45%). Patients with chronic obstructive pulmonary disease (COPD) had the highest number of DRPs and this was 15 cases. Twelve patients (20.69%), and 46 patients (79.31%) were found to be exposed to polypharmacy and not exposed to polypharmacy respectively. The p-value of 0.210 shown was at 0.05 level, meaning that there were no statistically significant differences between DRPs and length of hospital stay. The research concludes that the issue of DRPs is significant among patients taking antibiotics at tertiary care facilities. DRPs were selected and classified according to the PCNE classification system (version 9.1) using the type, cause, and outcomes.
Downloads
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Valikala Viswanath, Bhaskara Rao chavakala, M. Naveen, N. Supraja, S. Ibrahim, K. Yeswanth, D. Giri Raja Sekhar (Author)

This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
License Terms
This is an open-access article published in the International Journal of Research in Pharmaceutical Sciences and Technology (IJRPST) by Rubatosis Publications.
It is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, reproduction, and adaptation in any medium or format, provided the original author(s) and source are appropriately credited, a link to the license is provided, and any changes made are indicated.
To view a copy of this license, visit: https://creativecommons.org/licenses/by/4.0/
For any further queries or permissions beyond the scope of this license, please contact: editor@rubatosis.org





