Pharmacogenomics: bridging the gap between genetics and drug therapy
 DOI:
DOI:
https://doi.org/10.33974/s2vvt232
Keywords:
Pharmacogenomics, Precision medicine, Adverse drug reactions (ADRs), Genetic polymorphisms, Personalized therapy, Whole-genome sequencing, Cost-effectivenessAbstract
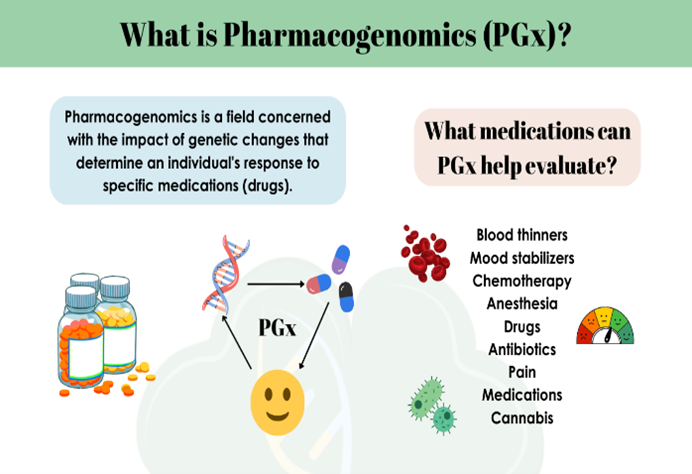
Pharmacogenomics, the integration of pharmacology and genomics, lies at the center of precision medicine by personalizing drug therapy based on genetics. 20–95% of drug response variability arises due to genetic variation, which influences drug metabolism, transport, target, and immune reactions. Adverse drug reactions (ADRs), a leading cause of hospitalization and death, make personalized prescribing critical. There are a number of major pharmacogenomic applications, including cardiology dosing of warfarin and clopidogrel, cancer treatment targeted to a specific cancer, psychiatric medication optimization, and prevention of hypersensitivity to drugs like abacavir and carbamazepine. CPIC and DPWG recommendations aid clinical application, with issues of test availability, provider education, ethical issues, and patient heterogeneity persisting. Pharmacogenomic testing may be costly initially but is cost-effective by avoiding hospitalization and maximizing treatment. Technological advances in AI, CRISPR, and whole-genome sequencing hold the potential for greater incorporation into medicine. Pharmacogenomics is a new model for therapeutics that allows for safer, more efficient, and individualized medicine, with global efforts in place to provide equitable access.
Downloads
References
Evans WE, McLeod HL. Pharmacogenomics—drug disposition, drug targets, and side effects. N Engl J Med. 2003;348(6):538–549. https://doi.org/10.1056/NEJMra020526
Roden DM, Wilke RA, Kroemer HK, Stein CM. Pharmacogenomics: the genetics of variable drug responses. Circulation. 2011;123(15):1661–1670. https://doi.org/10.1161/CIRCULATIONAHA.109.914820
Pirmohamed M, et al. Adverse drug reactions as cause of admission to hospital: prospective analy-sis. BMJ. 2004;329(7456):15–19. https://doi.org/10.1136/bmj.329.7456.15
Wiffen PJ, Gill M, Edwards JE. Adverse drug reac-tions in the elderly: a prospective-retrospective analysis. Drug Saf. 2002;25(8):571–580. https://doi.org/10.2165/00002018-200225080-00004
Carson PE, et al. Hemolytic anemia after prima-quine administration in G6PD-deficient patients. J Clin Invest. 1956;35(6):598–607. https://doi.org/10.1172/JCI103322
Kalow W, Gunn DR. The relation between dose of succinylcholine and duration of apnea in man. An-esthesiology. 1957;18(3):284–291. https://doi.org/10.1097/00000542-195705000-00004
Collins FS, Varmus H. A new initiative on precision medicine. N Engl J Med. 2015;372:793–795. https://doi.org/10.1056/NEJMp1500523
Zanger UM, Schwab M. Cytochrome P450 enzymes in drug metabolism. Pharmacol Ther. 2013;138(1):103–141. https://doi.org/10.1016/j.pharmthera.2012.12.007
Giacomini KM, et al. Membrane transporters in drug development. Nat Rev Drug Discov. 2010;9(3):215–236. https://doi.org/10.1038/nrd3028
Johnson JA, Cavallari LH. Warfarin pharmacogenet-ics. Trends Cardiovasc Med. 2015;25(1):33–41. https://doi.org/10.1016/j.tcm.2014.09.001
Mallal S, et al. Association between HLA-B*5701 and hypersensitivity to abacavir. Lancet. 2002;359(9308):727–732. https://doi.org/10.1016/S0140-6736(02)07873-6
Anderson JL, et al. Randomized trial of genotype-guided versus standard warfarin dosing. Circula-tion. 2007;116(22):2563–2570. https://doi.org/10.1161/CIRCULATIONAHA.107.737312
Mega JL, et al. Cytochrome P-450 polymorphisms and response to clopidogrel. N Engl J Med. 2009;360(4):354–362. https://doi.org/10.1056/NEJMoa0809171
Relling MV, et al. Pharmacogenetics of thiopurine methyltransferase. Clin Pharmacol Ther. 1999;66(6):506–516. https://doi.org/10.1016/S0009-9236(99)70008-2
Iyer L, et al. UGT1A1 polymorphism and irinotec-an-induced toxicity. J Clin Oncol. 1998;16(8):301–308. https://doi.org/10.1200/JCO.1998.16.8.301
Mok TS, et al. Gefitinib or carboplatin–paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361:947–957. https://doi.org/10.1056/NEJMoa0810699
Hicks JK, et al. Clinical Pharmacogenetics Imple-mentation Consortium guidelines for SSRIs. Clin Pharmacol Ther. 2015;98(2):127–134. https://doi.org/10.1002/cpt.147
Martin AM, et al. Predisposition to abacavir hyper-sensitivity by HLA-B*5701 screening. N Engl J Med. 2004;352(6):568–575. https://doi.org/10.1056/NEJMoa031093
Ge D, et al. Genetic variation in IL28B predicts hep-atitis C treatment response. Nature. 2009;461:399–401. https://doi.org/10.1038/nature08309
Relling MV, Klein TE. CPIC: guidelines for gene–drug pairs. Clin Pharmacol Ther. 2011;89(3):464–467. https://doi.org/10.1038/clpt.2010.279
Dunnenberger HM, et al. Implementing phar-macogenomics in practice: the US perspective. Clin Pharmacol Ther. 2015;98(5):468–471. https://doi.org/10.1002/cpt.190
You JH, et al. Pharmacoeconomic evaluation of warfarin genotyping. Pharmacogenomics. 2014;15(4):479–488. https://doi.org/10.2217/pgs.13.234
Verbelen M, et al. Cost-effectiveness of phar-macogenomics in primary care. Br J Clin Pharma-col. 2017;83(6):1147–1158. https://doi.org/10.1111/bcp.13216
Hughes DA, et al. Cost-effectiveness analysis of HLA-B*5701 testing. Pharmacogenetics. 2004;14(5):335–342. https://doi.org/10.1097/00008571-200405000-00007
Wetterstrand KA. DNA sequencing costs: NHGRI Genome Sequencing Program (GSP). Genome Res. 2025.
Stanek EJ, et al. Adoption of pharmacogenomic test-ing by US physicians. Clin Pharmacol Ther. 2012;91(3):450–458. https://doi.org/10.1038/clpt.2011.306
Vayena E, et al. Ethical frameworks for precision medicine. EMBO Mol Med. 2018;10(3):e7984. https://doi.org/10.15252/emmm.201707984
Popejoy AB, Fullerton SM. Genomics is failing on diversity. Nature. 2016;538(7624):161–164. https://doi.org/10.1038/538161a
Dunnenberger HM, et al. Preemptive clinical phar-macogenetics implementation. Am J Med Genet C Semin Med Genet. 2014;166C:45–55. https://doi.org/10.1002/ajmg.c.31391
Zhang Y, et al. AI in pharmacogenomics. Phar-macogenomics J. 2024;24(4):450–462. https://doi.org/10.1038/s41397-024-003xx
Doudna JA, Charpentier E. The new frontier of ge-nome engineering with CRISPR. Science. 2014;346(6213):1258096. https://doi.org/10.1126/science.1258096
All of Us Research Program Investigators. The “All of Us” Research Program. N Engl J Med. 2019;381(7):668–676. https://doi.org/10.1056/NEJMsr1809937
Downloads
Published
Issue
Section
License
Copyright (c) 2026 C. Mohana (Author)

This work is licensed under a Creative Commons Attribution 4.0 International License.
License Terms
This is an open-access article published in the International Journal of Research in Pharmaceutical Sciences and Technology (IJRPST) by Rubatosis Publications.
It is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, reproduction, and adaptation in any medium or format, provided the original author(s) and source are appropriately credited, a link to the license is provided, and any changes made are indicated.
To view a copy of this license, visit: https://creativecommons.org/licenses/by/4.0/
For any further queries or permissions beyond the scope of this license, please contact: editor@rubatosis.org





