Development and Validation of a UV-Visible Spectrophotometric Method for Quantitative Estimation of Empagliflozin in Tablet Dosage Forms
Keywords:
Empagliflozin , UV spectrophotometry , Method validation , Pharmaceutical analysis , Tablet dosage formAbstract
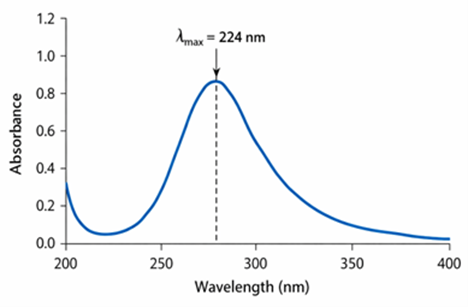
A simple, rapid, and economical UV-visible spectrophotometric method was developed and validated for the quantitative estimation of empagliflozin in tablet dosage forms. Empagliflozin exhibited maximum absorbance (λmax) at 224 nm in ethanol. The method showed good linearity in the concentration range of 2-12 µg/ml with a correlation coefficient (R2) of 0.999. The proposed method was validated according to the guidelines of the International Council for Harmonization of Technical Requirements of Pharmaceuticals for Human use Q2(R1) for linearity, accuracy, precision, robustness, limit of detection (LOD) and limit of quantification (LOQ). The percentage recovery ranged from 99.90 % to 100.07 %, indicating good accuracy of the method. Precision studies showed %RSD value below 2%, demonstrating acceptable repeatability. The LOD and LOQ were found to be 0.218 µg/ml and 0.661 µg/ml, respectively. The developed method was successfully applied for the assay of empagliflozin in marketed tablet formulation and was found to be suitable for routine quality control analysis due to its simplicit, accuracy and cost-effectiveness.
Downloads
References
Heise T, Seewaldt-Becker E, Macha S, Hantel S, Pin-netti S, Seman L, et al. Safety, tolerability, pharma-cokinetics and pharmacodynamics following 4 weeks’ treatment with empagliflozin once daily in patients with type 2 diabetes. Diabetes Obes Metab. 2013;15(7):613–21. https://doi.org/10.1111/dom.12073
Haering HU, Merker L, Seewaldt-Becker E, Weimer M, Meinicke T, Woerle HJ, et al. Empagliflozin as add-on to metformin plus sulfonylurea in patients with type 2 diabetes: a 24-week randomized dou-ble-blind placebo-controlled trial. Diabetes Care. 2013;36(11):3396–404. https://doi.org/10.2337/dc12-2673
Kovacs CS, Seshiah V, Swallow R, Jones R, Rattunde H, Woerle HJ, et al. Empagliflozin improves glyce-mic and weight control as add-on therapy to pioglitazone or pioglitazone plus metformin in pa-tients with type 2 diabetes. Diabetes Obes Metab. 2014;16(2):147–58. https://doi.org/10.1111/dom.12188
Roden M, Weng J, Eilbracht J, Delafont B, Kim G, Woerle HJ, et al. Empagliflozin monotherapy com-pared with sitagliptin in patients with type 2 diabe-tes: a randomized double-blind placebo-controlled phase 3 trial. Lancet Diabetes Endocrinol. 2013;1(3):208–19. https://doi.org/10.1016/S2213-8587(13)70084-6
Rosenstock J, Jelaska A, Frappin G, Salsali A, Kim G, Woerle HJ, et al. Improved glucose control with weight loss, lower insulin doses, and no increased hypoglycemia with empagliflozin added to titrated multiple daily injections of insulin in obese pa-tients with type 2 diabetes. Diabetes Care. 2014;37(7):1815–23. https://doi.org/10.2337/dc13-3055
Chilton R, Tikkanen I, Cannon CP, Crowe S, Woerle HJ, Broedl UC, et al. Effects of empagliflozin on blood pressure and markers of arterial stiffness and vascular resistance in patients with type 2 dia-betes. Diabetes Obes Metab. 2015;17(12):1180–93. https://doi.org/10.1111/dom.12572
Barnett AH, Mithal A, Manassie J, Jones R, Rattunde H, Woerle HJ, et al. Efficacy and safety of empagli-flozin added to existing antidiabetes treatment in patients with type 2 diabetes and chronic kidney disease. Lancet Diabetes Endocrinol. 2014;2(5):369–84. https://doi.org/10.1016/S2213-8587(13)70208-0
Tikkanen I, Narko K, Zeller C, Green A, Salsali A, Broedl UC, et al. Empagliflozin reduces blood pres-sure in patients with type 2 diabetes and hyperten-sion. Diabetes Care. 2015;38(3):420–8. https://doi.org/10.2337/dc14-1096
Padmaja N, Veerabhadram G. Method development and validation of RP-HPLC method for the estima-tion of empagliflozin in API. Int J Pharm Sci Res. 2016;7(2):457–64. https://doi.org/10.13040/IJPSR.0975-8232.7(2).457-64
Shyamala M, Soumika M, Sangeetha E, Mahender I. Method development and validation of empagli-flozin by RP-HPLC in bulk and pharmaceutical dosage form. Pharmanest. 2016;7(1):3040–4.
Shyamala K, Nirmala K, Mounika J, Nandini B. Val-idated stability-indicating RP-HPLC method for de-termination of empagliflozin. Der Pharm Lett. 2016;8(2):457–64.
Ansari MN. Chemical derivatization UV spectro-photometric method for detection of p-aminophenol and energy of activation approach to set degradation protocol for forced degradation studies. Int J Pharm Res Rev. 2016;5(2):1–12.
Behera S, Ghanty S, Ahmad F, Santra S, Banerjee S. UV-visible spectrophotometric method develop-ment and validation of assay of paracetamol tablet formulation. J Anal Bioanal Tech. 2012;3(6):151. https://doi.org/10.4172/2155-9872.1000151
International Council for Harmonisation. Valida-tion of analytical procedures: text and methodolo-gy Q2(R1). Geneva: ICH; 2005.
International Council for Harmonisation. Valida-tion of analytical procedures: methodology Q2B. Geneva: ICH; 1996.
Beckett AH, Stenlake JB. Practical pharmaceutical chemistry. 4th ed. New Delhi: CBS Publishers and Distributors; 2002.
Ramesh K, Silvestar AS, Shobana A, Surendhar P, Aafreen A, Kalaivani R, Nepolean R. Antenatal cor-ticosteroids for fetal lung maturation and preven-tion of respiratory distress syndrome: a systematic review. Int J Res Hosp Clin Pharm. 2026;8(1). https://doi.org/10.33974/ijrhcp.v8i1.248
Downloads
Published
Issue
Section
Categories
License
Copyright (c) 2026 N. Baskar, Nepolean R, Ramesh K, A. Adhithiyan, E. Maruthapandiyan, Abu Hanifa, F. Gibson Harrish (Author)

This work is licensed under a Creative Commons Attribution 4.0 International License.
License Terms
This is an open-access article published in the International Journal of Research in Pharmaceutical Sciences and Technology (IJRPST) by Rubatosis Publications.
It is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, reproduction, and adaptation in any medium or format, provided the original author(s) and source are appropriately credited, a link to the license is provided, and any changes made are indicated.
To view a copy of this license, visit: https://creativecommons.org/licenses/by/4.0/
For any further queries or permissions beyond the scope of this license, please contact: journals@rubatosis.org




